
"To measure absolute zero, you would actually need an infinitely accurate thermometer, and that's beyond our current measuring systems. "With current instruments, you couldn't tell whether it was zero or just a very, very small number," Foot said. Only a perfectly ordered, crystalline substance at absolute zero would exhibit no. 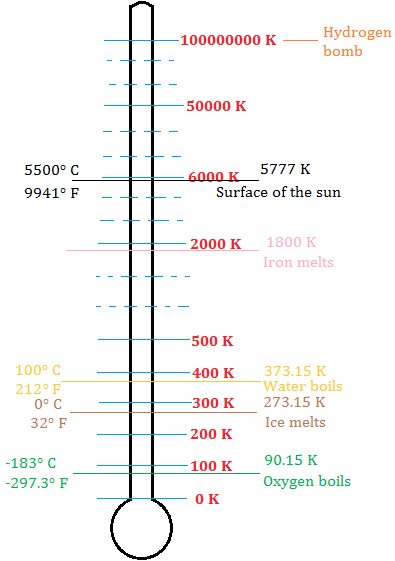
Vibrational, rotational, and translational motions of a carbon dioxide molecule are illustrated here. In fact, even if we were to reach absolute zero, we might completely miss it due to imprecise measuring techniques. The entropy of any perfectly ordered, crystalline substance at absolute zero is zero. Lower-temperature work is still at the research stage, and people are using these methods to test universal physical theories."Īt present, it's not possible to cool that final 38 trillionths of a degree - and several hurdles would have to be overcome for it to become a reality. "Laser-cooled atoms are already used in the atomic standards which define universal time (atomic clocks) and in quantum computers. "We're much more interested in these quantum effects than reaching absolute zero," he said. Where do electrons get energy to spin around an atom's nucleus?īut is there any point in trying to cool materials even further? Probably not, according to Foot. The third law of thermodynamics states that the entropy of a closed system at thermodynamic equilibrium approaches a constant value when its temperature approaches absolute zero. If there were a time warp, how would physicists find it? Absolute zero is the lowest possible temperature at which molecules that make up a substance move as slow as they can. Entropy is one of the most fundamental concepts of physical science, with far-reaching consequences ranging from cosmology to chemistry. In this type of experiment, known as magnetic trap cooling, the gaseous particles reached an incredible 38 picokelvin - 38 trillionths of a degree Celsius above absolute zero and well within the range to begin observing quantum effects in gases. The absolute entropy of a pure substance at a given temperature is the sum of all the entropy it would acquire on warming from absolute zero (where S0) to the particular temperature. 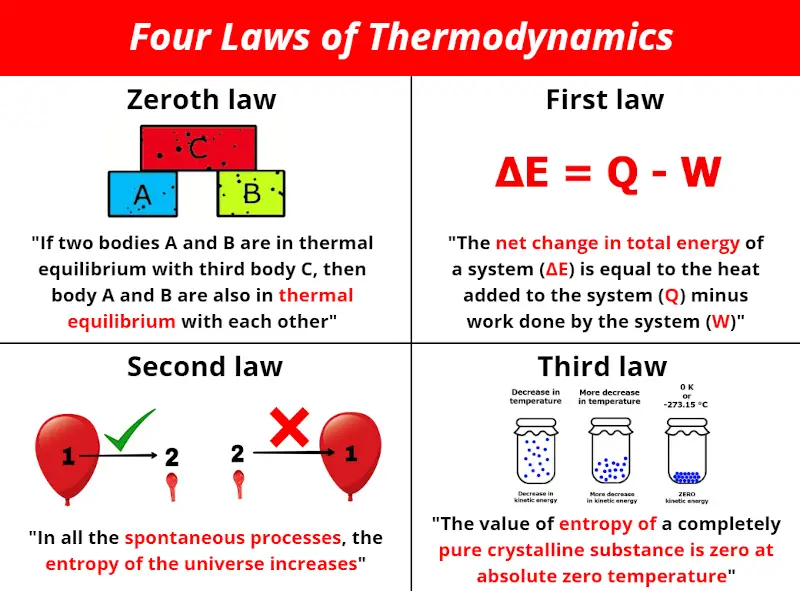
The team dropped magnetized gas atoms down a 400-foot (120 meters) tower, constantly switching the magnetic field on and off to slow the particles to almost a complete standstill. The lowest temperature ever recorded in a lab was achieved by a group in Germany in 2021. " to see quantum behavior in solids and liquids but for the gases we study, we need 10s of nano-kelvin temperatures to get these quantum effects." "The light exerts a force on the atoms which slows them down to reasonably cold temperatures, around 1 kelvin (minus 272.15 C or minus 457.87 F)," said Christopher Foot, an ultracold physicist at the University of Oxford. (Image credit: dra_schwartz via Getty Images)Įarly ultracold experiments in the 1990s used a technique known as laser cooling to begin probing these effects. Try as they might, scientists have never knowingly reached absolute zero during an experiment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
